Abstract
Introduction: There are no prospective randomized data supporting the use of upfront consolidative autologous stem cell transplant (ASCT) in newly diagnosed mantle cell lymphoma (MCL) patients (pts). The aim of this population-based study in MCL pts is to better define the impact of frontline ASCT on overall survival (OS), and to identify factors influencing treatment selection.
Methods: Using the National Cancer Data Base (NCDB), we identified pts with newly diagnosed MCL treated from 2004-2012 with multi-agent chemo alone or in combination with consolidative ASCT as part of first line treatment. Univariable and multivariable Cox proportional hazards regression analyses were used to identify predictors for treatment selection and OS, including age, gender, race, stage, Charlson-Deyo comorbidity index (CDCI), income, education, insurance, treatment facility type, distance to treatment facility and year of diagnosis. Income was recorded as quartiles of median annual household income and education as percentage of persons with less than high school education in pts' residence census. Including these same factors, a propensity score matched analysis (PSMA) was performed to further account for confounding variables.
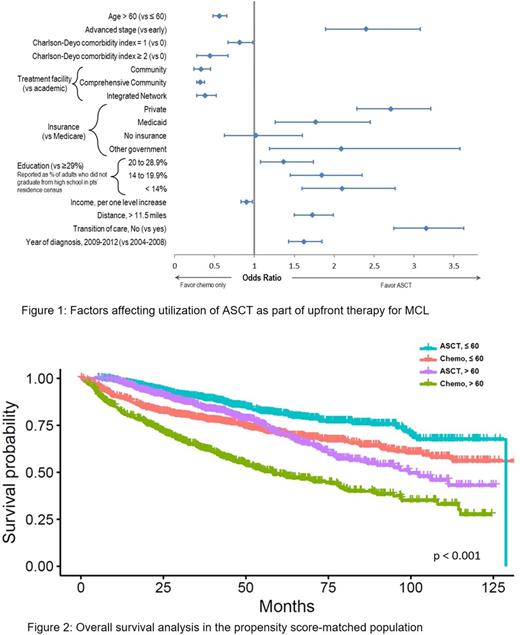
Results: We identified 16,035 MCL pts with known survival times. The following groups were excluded: no chemo (n=3,193), chemo status/type unknown (n=1,056), chemo contraindicated (n=199), single-agent chemo (n=983), received allogeneic SCT (n=79) or ASCT data missing (n=235). The final analysis included 10,290 pts. Median age was 65 years (range 20-90); 66% were > 60 years, 73% male, 93% Caucasian, and 78% had stage III-IV. CDCI was 0 in 79%, 1 in 16% and ≥2 in 5%. Forty percent of pts were treated at academic institutions and 49% travelled >11.5 miles to receive treatment. Medicare was the most common insurance type (48%), followed by private (43%) and Medicaid (4%). Median follow up was 34 months (interquartile range (IQR) 16-60). Median OS for the entire cohort was 64 months; 5- and 10-year OS rates were 52% and 33%, respectively. Seventeen percent of the pts (n=1774) underwent upfront ASCT, and there was a trend of increased use from 2004-2012 (11.9% in 2004 and 21.2% in 2012) that resulted in an annual increase of 1.2% (p<0.001). 31% of pts ≤60 and 25% of pts ≤70 years received ASCT. For the ASCT group, median age was 58 years (IQR 52-64; range 23-77); 89 pts (5%) were ≥70 years of age. Predictors for ASCT use included (p<0.001 for each): stage III-IV (odds ratio (OR) 2.4), having private insurance (OR 2.71), higher education (OR 2.1), no transition of care (diagnosis and treatment done at the same facility) (OR 3.15), longer distance (> 11.5 miles) to treatment facility (OR 1.73) and year of diagnosis ≥2009 (OR 1.62) (Figure 1). Median follow up was 44 months (IQR 28-69) for ASCT group and 31 months (IQR 14-58) for chemo only group. Five and 10-year OS rates for ASCT and chemo group were 77%, 57%, 47% and 28%, respectively. After adjusting for other covariates, the use of ASCT was associated with superior OS (HR: 0.46; 95% CI 0.41-0.52; p<0.001). Other factors associated with superior OS were: female gender (HR 0.92, p=0.01), income (HR 0.92, p<0.001), private insurance (HR 0.71, p<0.001), no transition of care (HR 0.82, p<0.001) and diagnosis year ≥2009 (HR 0.91, p=0.005). Predictors for inferior OS were: age >60 (HR 1.62, p<0.001), African American race (HR 1.23, p=0.009), stage III-IV (HR 1.58, p<0.001) and CDCI ≥2 (HR 2.0, p<0.001). Using a subset of 1582 pts treated with ASCT and 1582 matched controls treated with chemo only, PSMA confirmed the OS benefit associated with ASCT (HR 0.5; CI 0.44-0.57, p<0.001). This OS benefit was observed in both younger (≤60 years: HR 0.44, CI 0.37-0.53, p<0.001) and older pts (>60 years: HR 0.59, CI 0.46-0.68; p<0.001) (Figure 2).
Conclusion: In this large MCL cohort, the use of upfront consolidative ASCT was associated with superior OS when compared to chemo alone. Less than one third of young MCL pts treated with multi-agent chemo received consolidative ASCT, and this was influenced by sociodemographic factors with patterns favoring pts with higher education and private insurance. While one caveat in interpreting this retrospective study is that ASCT may have been preferentially applied to pts who responded to induction therapy, these data suggest clinical efficacy of ASCT in MCL. Prospective randomized trials are needed for further validation.
Majhail: Sanofi: Honoraria; Anthem, Inc.: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.